Research interest
Research Interest
Effective brain function depends upon accurate information transfer from one region of the brain to another through synaptic connections, and misconnections frequently lead to the development of neurological disorders. Connectivity within neural networks can be changed by experience and external stimuli, thereby allowing neurons to adjust their respond new environments. Although macroscopic circuit changes caused by altered sensory experience have been described, the underlying changes in distribution and strength of synaptic connections at the level of synapses remain unknown. This lack of knowledge is problematic. because, without it, macroscopic misconnections cannot be understood and this limits our ability to comprehend and treat the underlying pathological causes of many neurological diseases. The long term goal of the our lab is to decipher how neocortical neurons in healthy and pathological brain integrate and process information and to establish how experience-dependent plasticity shapes synaptic connections. To achieve the objective, we have successfully adapted two independent, complimentary methods to identify distinct synaptic populations; array tomography with correlative EM and Channelrhodopsin-2 (ChR2) aided subcellular synaptic input mapping. Array tomography is a new imaging technique that allows combination of high isotropic resolution and large volumetric field of view with discrimination of multiple molecular markers. This method is based on ultrathin serial sectioning of resin-embedded tissue and computational reconstruction of a three dimensional volume of tissue. With this method, we can reconstruct multiple whole cortical neurons and detect synaptic connections on the neurons that have been genetically labeled in volumes of ~mm3 of neocortex. ChR2, a light-gated cation channel, is delivered to the source of synaptic input, allowing reliable and selective photostimulation of the input synapses. Subcellular location of these synapses on cortical neurons can be detected by 2-PLSM Ca2+ imaging of cortical neurons.
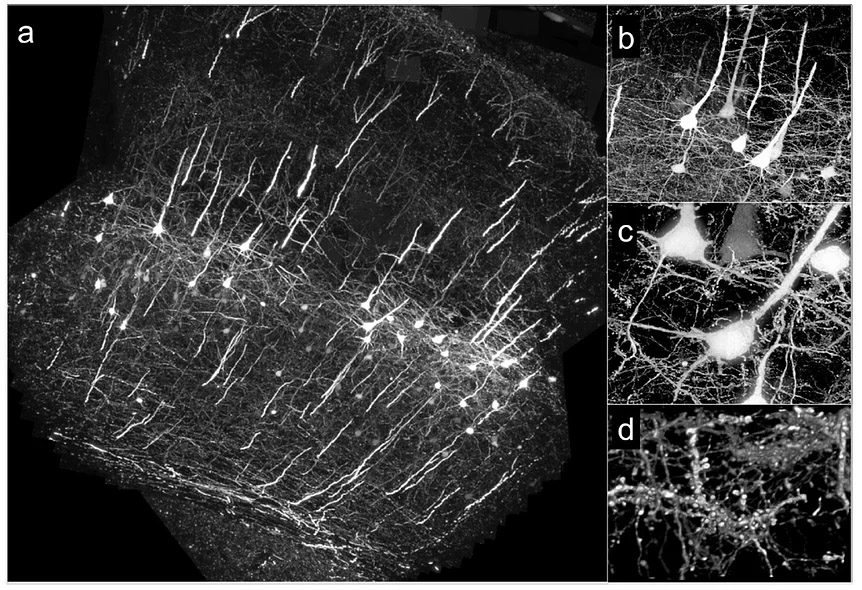
For Large-Scale Array Tomography, we prepared 1074 serial ribbon sections (thickness, 200 nm) of barrel cortex and immunostained each as above. Imaging was carried out on an automated inverted fluorescence microscope using a 100x, 1.45 N.A.objective and custom autofocussing (see our previous report). Each section (area, 1.00 × 0.83 mm^2) was imaged as a series of 240 overlapping tiles for a total of 257,760 images
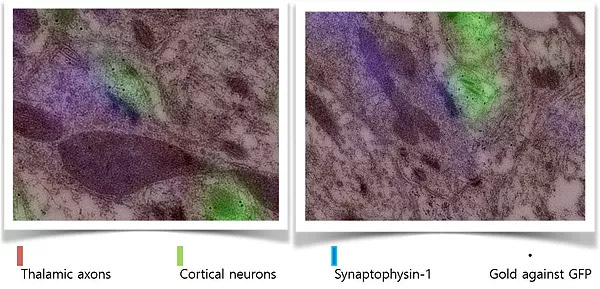
We verified the ability of array tomography to identify synapses using parallel electron microscopic analysis of TC synapses: Majority of thalamocortical synapses were correctly identified.
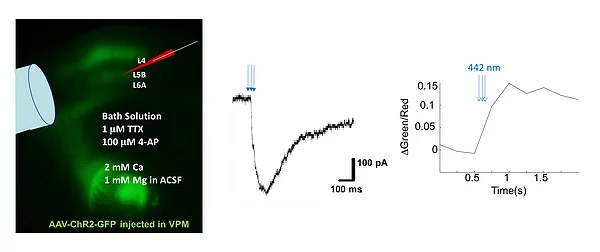
Synapses originated from particular group of neurons can also be identified by the combination of optogenetic stimulation and Ca imaging on acute brain slices. This method allows us to examine differences in physiological properties of synapses with different origins


